Abstract
OBJECTIVE: To assess whether hippocampal sclerosis (HS) severity is mirrored at the level of large-scale networks.
METHODS: We studied preoperative high-resolution anatomical and diffusion-weighted MRI of 44 temporal lobe epilepsy (TLE) patients with histopathologic diagnosis of HS (n = 25; TLE-HS) and isolated gliosis (n = 19; TLE-G) and 25 healthy controls. Hippocampal measurements included surface-based subfield mapping of atrophy and T2 hyperintensity indexing cell loss and gliosis, respectively. Whole-brain connectomes were generated via diffusion tractography and examined using graph theory along with a novel network control theory paradigm that simulates functional dynamics from structural network data.
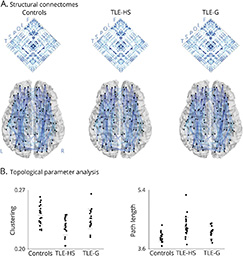
RESULTS: Compared to controls, we observed markedly increased path length and decreased clustering in TLE-HS compared to controls, indicating lower global and local network efficiency, while TLE-G showed only subtle alterations. Similarly, network controllability was lower in TLE-HS only, suggesting limited range of functional dynamics. Hippocampal imaging markers were positively associated with macroscale network alterations, particularly in ipsilateral CA1-3. Systematic assessment across several networks revealed maximal changes in the hippocampal circuity. Findings were consistent when correcting for cortical thickness, suggesting independence from gray matter atrophy.
CONCLUSIONS: Severe HS is associated with marked remodeling of connectome topology and structurally governed functional dynamics in TLE, as opposed to isolated gliosis, which has negligible effects. Cell loss, particularly in CA1-3, may exert a cascading effect on brain-wide connectomes, underlining coupled disease processes across multiple scales.